LATEST NEWS

Enrolment reaches midway point in trial of Early Bird bleed monitoring system
Saranas has announced that it has reached the midpoint for enrolment in SAFE-MCS, a multicentre, single arm, open-label clinical trial evaluating the safety of complex high-risk percutaneous coronary intervention (PCI) using mechanical circulatory support (MCS) and surveillance with the Early Bird bleed monitoring system.

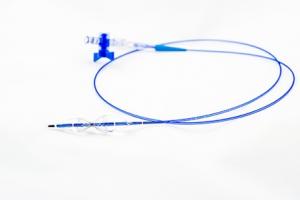
Vesteck completes first-in-human use of several Suture-Tight devices
Vesteck scientific advisory board members along with key opinion leaders from around the world have successfully completed the first three in-human cases with the Suture-Tight catheter, a press release reports.