Researchers have detailed the use of an artificial intelligence-enabled tool for atherosclerotic plaque quantification from coronary CT angiography (CCTA), which they claim may make it easier to predict if a person will have a heart attack.
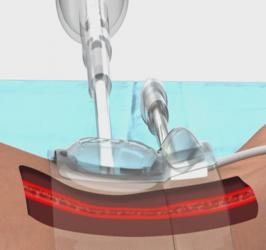
Cardiawave has announced the success of its first-in-human I (FIH I) safety and feasibility clinical trial for Valvosoft its breakthrough technology that allows for the remote application of a focused therapeutic ultrasound beam to restore valve function in patients with aortic stenosis (AS).
A consensus document outlining design principles and outcome definitions for device-based therapies for hypertension has beenpublished in the journal Circulation.
Four radial cases using the PH Band System (Medical Ingenuities)—a novel medical device that is used to close the arteriotomy of a radial artery following a Percutaneous Coronary Intervention (PCI) procedure—have been completed at the Lakewood Ranch Medical Center (Bradenton, USA).
Only 76% of heart attack patients with dyspnoea or fatigue as their main symptom are alive at one year compared to 94% of those with chest pain as the predominant feature. That is the finding of research presented at the European Society of Cardiology (ESC) Acute CardioVascular Care 2022 virtual meeting (18–19 March, online).
Xeltis has announced promising initial outcomes from two key vascular trials involving its restorative graft technologies.
A new strategy using telehealth to monitor blood pressure at home for several months immediately after a stroke had a positive impact on patient engagement and blood pressure control among people who live in historically under-resourced communities, according to preliminary research presented at this year’s International Stroke Conference (ISC 2022; 9–11 February, New Orleans, USA).
Four-year data from the LRT—Low Risk TAVR—trial show that transcatheter aortic valve implantation (TAVI) in low-risk patients with symptomatic severe aortic stenosis yields low adverse event rates and excellent haemodynamics, according to LRT principal investigator Ron Waksman (Medstar Washington Hospital Center, Washington DC, USA).
Levels of high-sensitivity troponin I after cardiac surgery that were associated with an increased risk of death within 30 days were substantially higher than levels currently recommended to define clinically important periprocedural myocardial injury, a study in the New England Journal of Medicine has concluded.
Biotronik has announced final enrolment in the BIOMAG-I clinical trial by Michael Haude (Rheinland Klinikum, Neuss, Germany) the study’s coordinating clinical investigator and the principal investigator Javier Escaned (Hospital Universitario Clínico San Carlos, Madrid, Spain. The prospective single-arm study assesses the safety and clinical performance of Dreams 3G resorbable magnesium scaffold (RMS).
Women remain underrepresented in leading cardiovascular clinical trials, corresponding with underrepresentation and a lack of gender diversity among presenters at scientific meetings, according to a review published in the Journal of the American College of Cardiology (JACC).
Final five-year results from the BIOFLOW-V trial, presented during a late-breaking trial session at CRT 2022 (27 February–1 March, Washington DC, USA), affirm the durability of longer-term outcomes with the ultrathin Orsiro coronary drug-eluting stent (DES) compared to Xience (Abbott).

Performance diagnostique de l’interféron gamma dans l’identification de l’origine tuberculeuse des pleurésies exsudatives

A Mixed Phenotype of Airway Wall Thickening and Emphysema Is Associated with Dyspnea and Hospitalization for Chronic Obstructive Pulmonary Disease.

Radiological Approach to Asthma and COPD-The Role of Computed Tomography.

Significant annual cost savings found with UrgoStart in UK and Germany

Thrombolex announces 510(k) clearance of Bashir catheter systems for thromboembolic disorders
Phone: (028) 3981 2678
Mobile: 0903 839 878 - 0909 384 389