A retrospective observational study of patients undergoing cardiac surgery in the UK between March and April 2020, at the height of the first wave of the COVID-19 pandemic, has found that those who were diagnosed with COVID-19 at the time of surgery had greater risk of death than those without COVID-19.
Acutus Medical has announced US Food and Drug Administration (FDA) clearance of the AcQCross family of universal transseptal crossing devices. The transseptal puncture system is specifically engineered to pair with Acutus’ own suite of sheaths and with sheaths sold by other manufacturers.
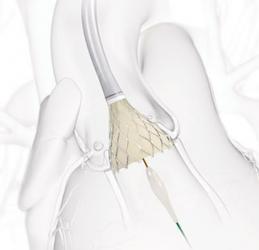
The first procedure has been completed in SMART (Small Annuli Randomized to Evolut or Sapien) post-market trial, a randomised, head-to-head study comparing two transcatheter aortic valve implantation (TAVI) systems in patients with severe symptomatic aortic stenosis (AS).
Abbott has received CE mark for its next-generation TriClip transcatheter tricuspid valve repair system, the first-of-its-kind minimally invasive tricuspid heart valve repair device available in Europe to treat tricuspid regurgitation (TR).
Surmodics recently announced the successful first uses in patients for two devices within its Sublime radial access platform: the Sublime radial access guide sheath and Sublime radial access .014 RX percutaneous transluminal angioplasty (PTA) dilatation catheter.
While there is large variability based on the site of insertion, patient characteristics and previous accesses, fluoroscopically-guided insertion of tunnelled central venous catheters (td-CVC) for dialysis can be considered a “low exposure” procedure, according to a report.
“The available data suggest that exercise training improves de-oxygenation and re-oxygenation patterns, as measured with near infrared spectroscopy (NIRS), in patients with lower extremity arterial disease (LEAD),” conclude Nils Cornelis, Panagiotis Chatzinikolaou (both KU Leuven, Leuven, Belgium) and colleagues in the European Journal of Vascular and Endovascular Surgery (EJVES). They add that their systematic review highlights the heterogeneity of NIRS application methods, outcome variables, and analysis in the literature. However, the authors stress that “more robust” studies are needed to confirm NIRS as a valuable diagnostic tool in patients with LEAD.
Abbott has received CE mark for its next-generation TriClip transcatheter tricuspid valve repair system, the first-of-its-kind minimally invasive tricuspid heart valve repair device available in Europe to treat tricuspid regurgitation (TR).
Abbott has announced that its Xience stent has received CE mark in Europe for shorter duration of dual anti-platelet therapy (DAPT)—including for 28 days—for patients with high bleeding risk (HBR).
ClearFlow has announced clinical trial results published in theJournal of Cardiothoracic Surgeryevaluating the use of the PleuraFlow active clearance technology (ACT) system.
In a large, multicentre study, women undergoing fenestrated or branched endovascular aneurysm repair (F/BEVAR) for thoracoabdominal aortic aneurysms (TAAA) demonstrated metrics of increased complexity and had a lower level of technical success. However, women had similar 30-day mortality and one-year outcomes to men, with the exception of an increased incidence of sac expansion. “These data demonstrate that F/BEVAR is safe and effective among women and men”, write Natasha I Edman (University of Washington Medical Center, Seattle, USA) and colleagues in the Journal of Vascular Surgery (JVS). However, they stress that “further efforts to improve outcome parity are indicated”.
Today, W L Gore & Associates (Gore)announced the first use of the US Food and Drug Administration (FDA)-approved Gore Excluder conformable abdominal aortic aneurysm (AAA) endoprothesis with Active Control system in cases outside of clinical trials. Cases were successfully performed by Robert Rhee with Mahmoud Almadani at Maimonides Medical Center in New York, USA, and by Gustavo Oderich with Naveed Saqib at UTHealth in Houston, USA.

Performance diagnostique de l’interféron gamma dans l’identification de l’origine tuberculeuse des pleurésies exsudatives

A Mixed Phenotype of Airway Wall Thickening and Emphysema Is Associated with Dyspnea and Hospitalization for Chronic Obstructive Pulmonary Disease.

Radiological Approach to Asthma and COPD-The Role of Computed Tomography.

Significant annual cost savings found with UrgoStart in UK and Germany

Thrombolex announces 510(k) clearance of Bashir catheter systems for thromboembolic disorders
Phone: (028) 3981 2678
Mobile: 0903 839 878 - 0909 384 389